Discover the difference of sbeadex technology
Experience sbeadex nucleic acid purification kits, where advanced technology meets seamless user-friendly operation. All of our sbeadex nucleic acid purification kits leverage the power of magnetic bead technology by utilising superparamagnetic particles coated with sbeadex surface chemistry. A novel binding mechanism ensures high-quality DNA or RNA, removing impurities from your sample matrix, and preparing it for PCR, sequencing and other downstream applications. Our kits are optimised for high-throughput automation and are compatible with popular robotic platforms including KingFisher, Hamilton, Tecan, Beckman-Coulter, and our fully automated nucleic acid extraction platform oKtopure™.
Explore our sbeadex nucleic acid purification kits

Save time and streamline your workflow with our new Lightning Nucleic Acid Purification Kits
Our new sbeadex™ Lightning product line provides a revolutionary approach to nucleic acid isolation with a superfast and simple 3-step protocol, typically purifying nucleic acids in just 5 minutes from a variety of lysed plant and animal sample materials. Using sbeadex Lightning, a minimum of 4-fold increases in sample throughput can be achieved compared to competitor chemistries. Reductions in liquid waste (by an average of 60%) and plastic waste (by an average of 50%) are achievable compared to magnetic bead-based and spin column-based competitor kits whilst boasting the simplest and fastest nucleic acid purification workflow in the market.
sbeadex Lightning at a glance
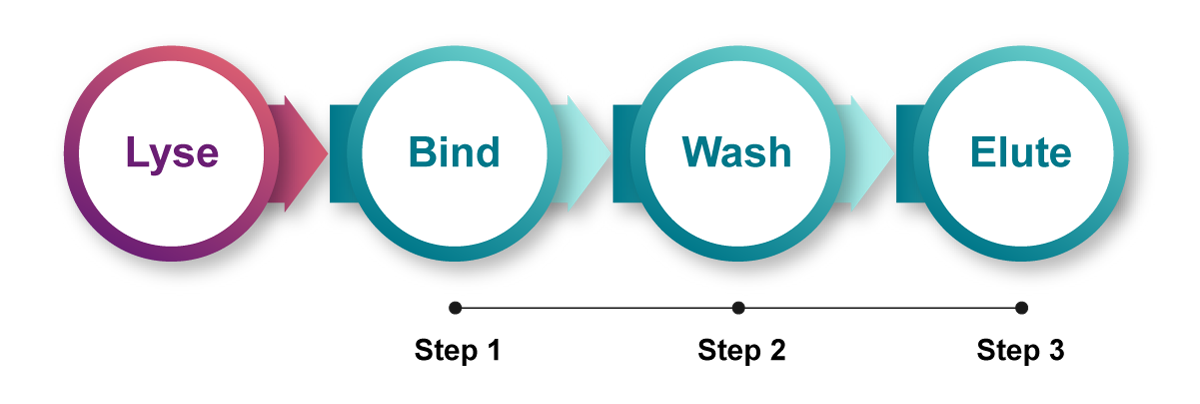
sbeadex Lighting has brought game-changing innovative solutions to traditional nucleic acid purification. Our cutting-edge technology delivers high-quality DNA in high yield, all within a simple, ultrafast 3-step/5-minute protocol designed to save time and costs. With fewer protocol steps, the error probability is lower, ensuring reliable and consistent results for every purification.

Transforming the traditional, time-consuming steps of nucleic acid purification – sbeadex Lightning revolutionises your workflow, reducing it to just 3 steps and 5 minutes
Powered by superparamagnetic particles and a novel one-step binding mechanism, sbeadex Lighting efficiently binds and purifies nucleic acids. Combined with a single water washing step, this unique process effectively removes impurities and potential inhibitors of enzymatic reactions, preparing nucleic acids for a wide range of downstream applications, including PCR, RT-PCR, sequencing/NGS, and restriction analysis.
- Superfast, simplified, and convenient, our 3-step/5-minute protocol saves time and money, and is easily adaptable to high-throughput automation
- Highly purified DNA suitable for many downstream applications/li>
- No potential PCR inhibitors (e.g. organic solvents/ethanol, salts) in final wash step and eluate
- Environmentally friendly kits and reagents: reduces plastics, packaging, shipping costs and hazardous waste
- Ready-to-use reagents with high stability at room temperature
- Compatible with most popular robotic platforms (e.g. KingFisher, Hamilton, Tecan, Beckman-Coulter, or our high-throughput oKtopure™ instrument)
sbeadex™ Lightning nucleic acid purification chemistry
Our new sbeadex Lightning technology offers all the advantages of the original sbeadex – but revolutionises the rather time-consuming, multi-step workflow of current nucleic acid purification methods. sbeadex Lightning uses superparamagnetic particles and a novel one-step binding mechanism to bind and purify nucleic acids. Combined with a single water washing step, this unique process removes impurities and potential inhibitors of enzymatic reactions very effectively, preparing nucleic acids for use in downstream applications including PCR and RT-PCR, sequencing, NGS and restriction analysis.
The very simple purification workflow involves only 3 steps and typically requires just 5 minutes from lysate to DNA while delivering high yields of pure DNA.
It therefore dramatically increases potential sample throughput per day – either through manual or automated nucleic acid purification – saving time and laboratory costs. Fewer protocol steps also mean fewer buffers, fewer plastic consumables and reduced liquid waste which helps to protect the environment (table 1).
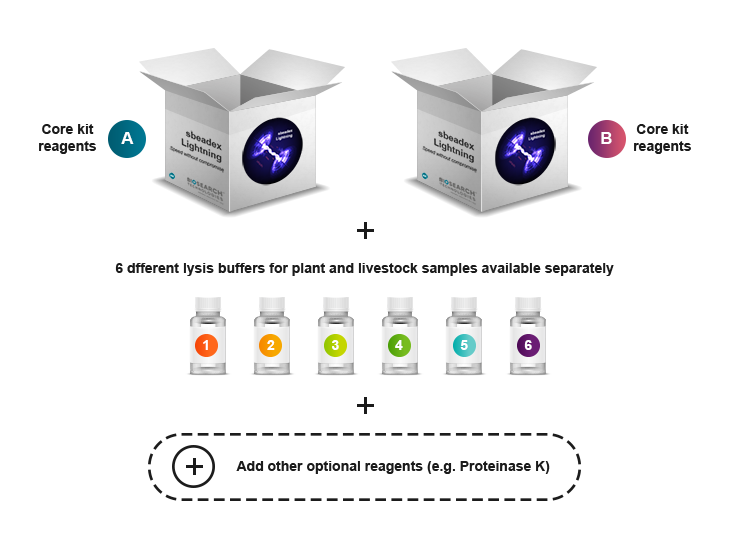
We recommend starting with our comprehensive starter kit to evaluate performance and optimise workflow before progressing to one of our core kits. The sbeadex Lightning Starter Kit is designed as an essential first step, offering the opportunity to test and determine the optimal core kit and lysis conditions. We offer two versions of the sbeadex Lightning core kits (Kit A or Kit B), each optimised under different conditions and for specific sample types. These kits include a sbeadex Lightning binding buffer, sbeadex particle suspension, and elution buffer (AMP).
The reagents of the sbeadex Lightning chemistry are not only reduced in volume but are also less hazardous than common reagents used in classical magnetic bead, spin column or solution-based workflows. Thus, our new sbeadex Lightning is not only faster, simpler and more convenient – but also more environmentally friendly. Compared to the classical magnetic bead or spin-column based technologies, sbeadex Lightning shows comparable results with regard to DNA purity, quality and yields – but is significantly faster, simpler and cost-saving (see tables 1-4).
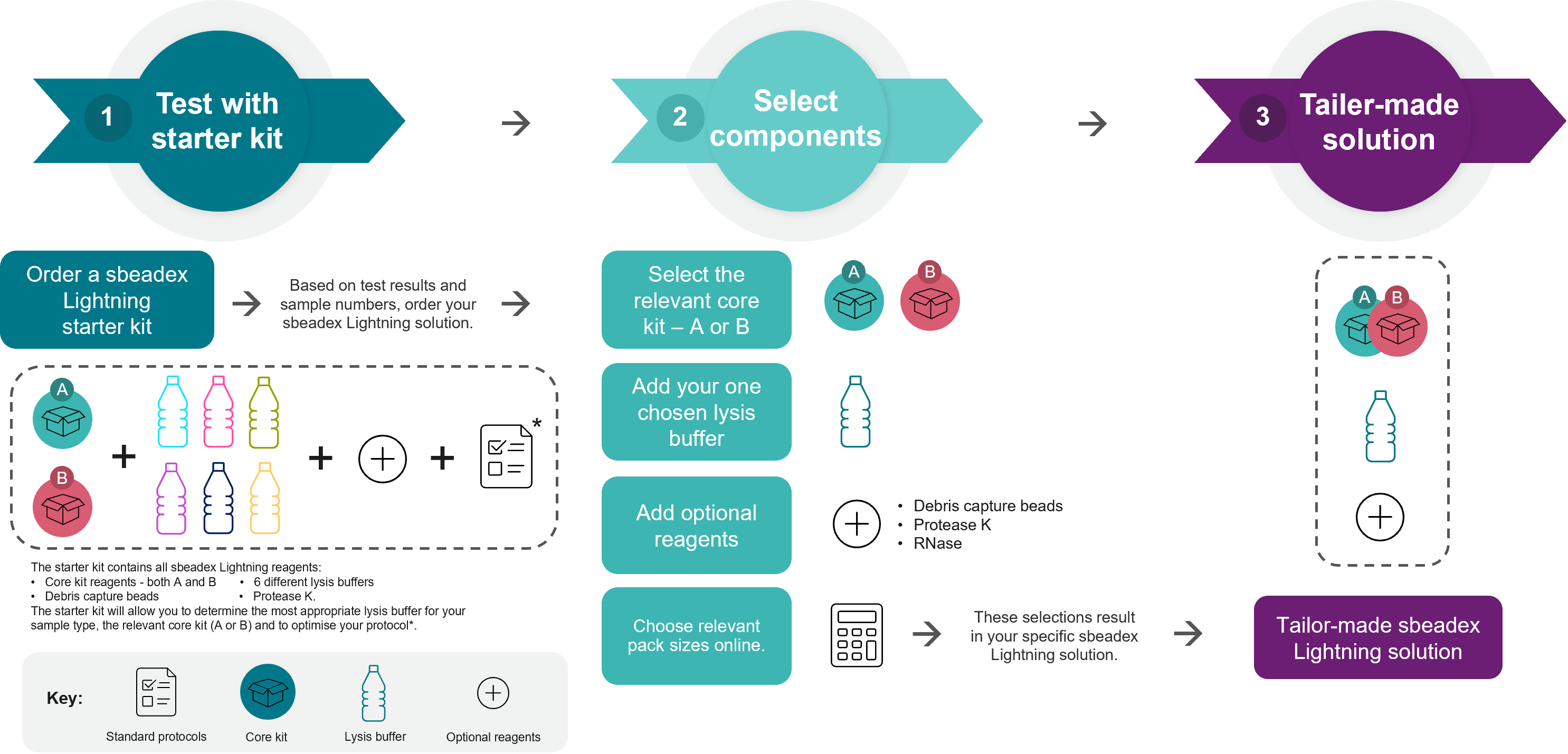
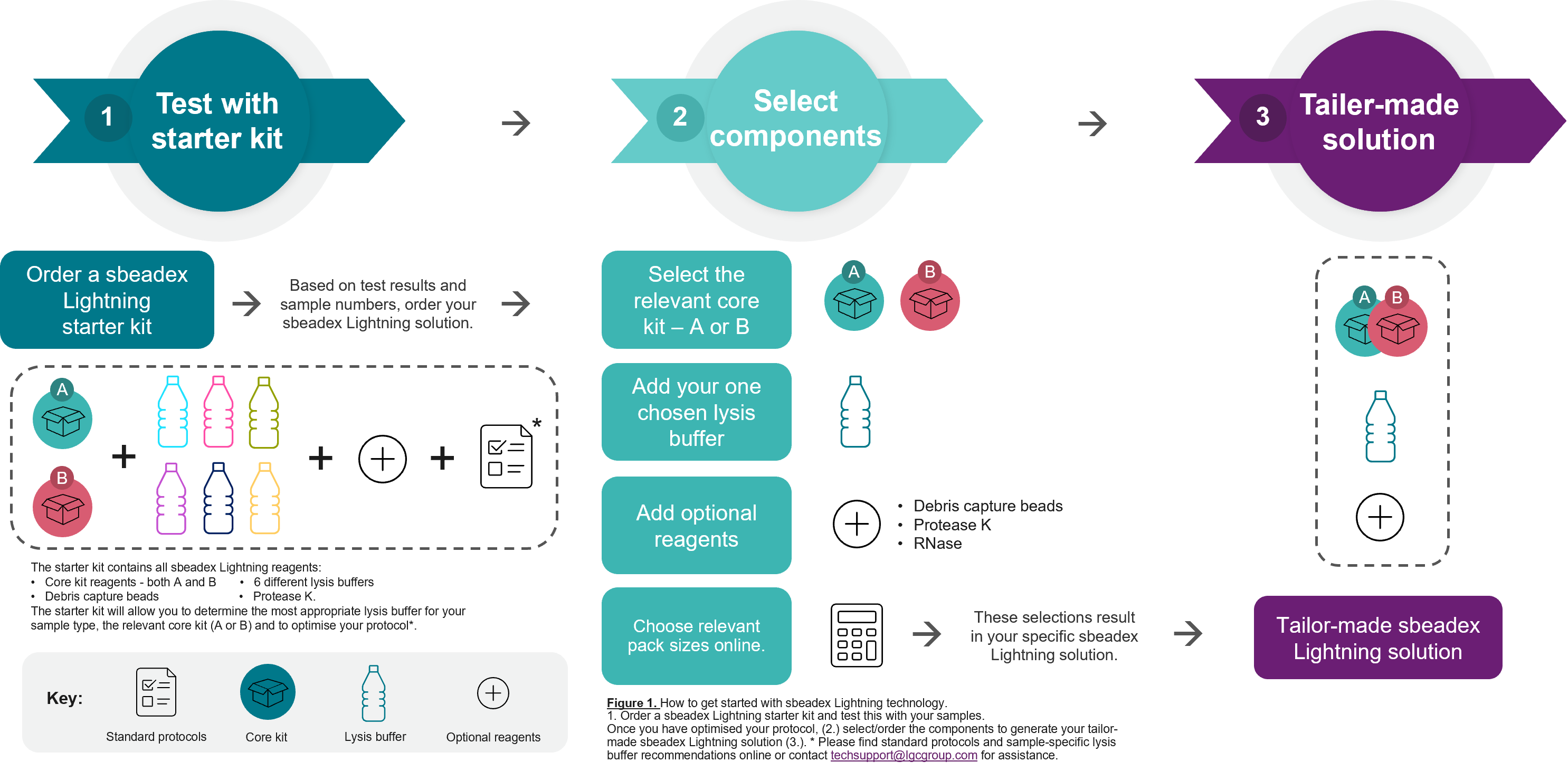
Figure 1. How to get started with sbeadex Lightning technology.
1. Order a sbeadex Lightning Starter Kit and test this with your samples. Once you have optimised your protocol, (2.) select/order the components to generate your tailor-made sbeadex Lightning solution (3.). * Please find standard protocols and sample-specific lysis buffer recommendations online or contact techsupport@lgcgroup.com for assistance.
The sbeadex Lightning chemistry comes as a modular system to enable you to compile an optimal kit tailored to your specific needs – see figure 1 below. The technology involves a core kit, either Kit A or Kit B, consisting of binding buffer, sbeadex particle suspension and elution buffer. In addition, there is a range of individual components (e.g. lysis buffers, Proteinase K solution, debris capture beads) that can be purchased separately. To help with optimisation of the lysis conditions, we also provide a sbeadex Lightning Starter Kit that includes both core kits, six different lysis buffers (PN, PVP, UR, BL, H, LI), Protease K solution and debris capture beads.
There are two different sbeadex Lightning core kits:
- Core Kit A is suitable for most sample types and is recommended for most plant samples (see tables 1 and 2 in technology and application data section).
- Core Kit B is suitable for specific sample types, especially mammalian tissue samples.
This game-changing innovation requires only a binding buffer, our proprietary sbeadex particles and an elution buffer to purify DNA quickly from a lysed sample. The (optional) wash step is performed with water, preventing organic/alcoholic traces in the eluate that might inhibit enzymatic downstream applications. This reduction in required buffers also saves plastic bottles and packaging material as well as storage space and shipping costs which additionally contributes to the environmental sustainability of our products.
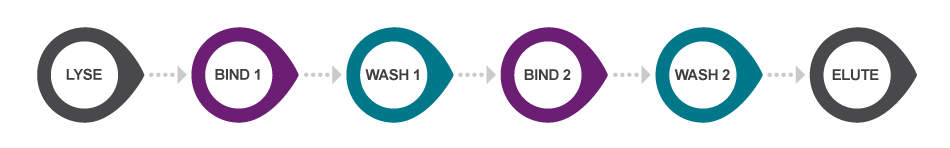
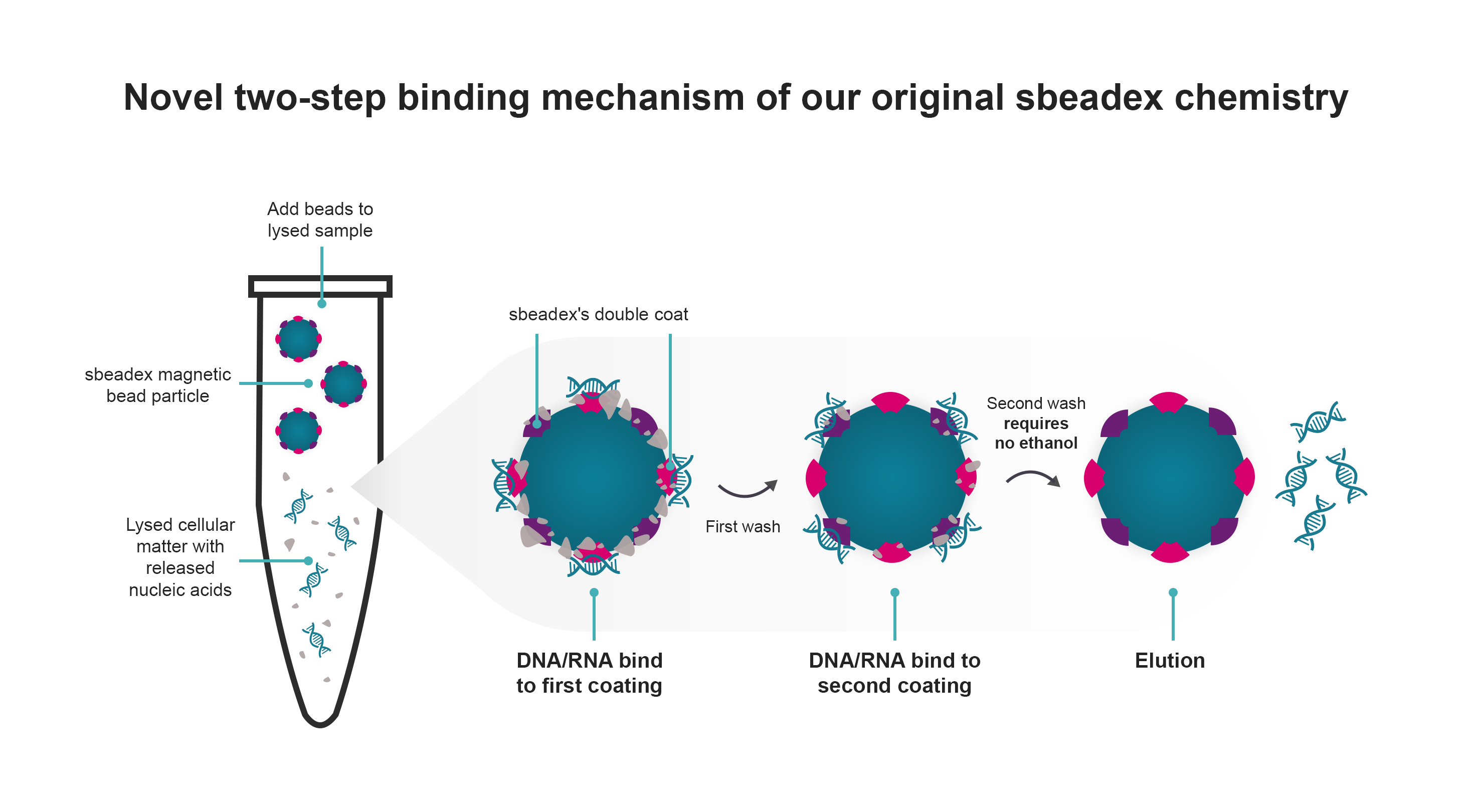
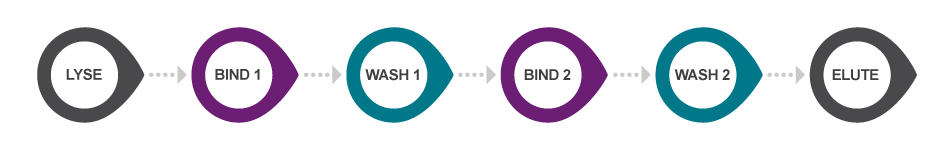
Our original sbeadex chemistry utilises a novel two-step binding mechanism to bind and purify nucleic acids, in a 5-step protocol (post-lysis) that requires approximately 45 minutes processing time. The sbeadex beads allow efficient and high-quality purification of both small and large nucleic acid fragments without centrifugation steps. The beads are double-coated, resulting in a unique dual binding protocol that allows elution of ultrapure nucleic acids into water, significantly reducing solvent carryover that might affect downstream applications.
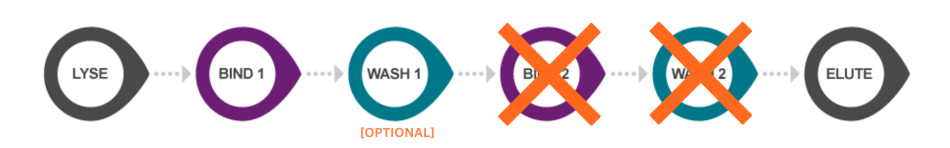
sbeadex Lightning nucleic acid purification protocol. By utilising a revolutionary novel one-step binding mechanism, sbeadex Lightning reduces the traditional magnetic bead-based nucleic acid purification workflow to a 3-step protocol (post-lysis) that typically requires just 5 minutes processing time.
Applications
- DNA purification for many downstream applications such as PCR/qPCR, RT-PCR or NGS
- Applicable to a wide range of sample types including plant and animal tissues (tables 1 and 2)
| Plant tissue | ||
|---|---|---|
| leaf | ||
| cucumber leaves | barley leaves | carrot leaves |
| brassica leaves | corn leaves | melon leaves |
| tomato leaves | potato leaves | cannabis leaves |
| pepper leaves | pea leaves | oat leaves |
| wheat leaves | parsley | |
| seed | ||
| corn seed | canola seed | soy seed |
| sunflower seed | ||
| fruit | ||
| blackberry | blueberry | potato |
Table 1: Plant sample types successfully tested using the sbeadex Lightning chemistry for DNA purification.
| Animal tissue | |
|---|---|
| bovine ear punch | mouse ears |
| bovine meat | mouse tail |
| bovine hairs | Chicken (wing) |
| pig ear punches | Salmon (fin) |
| Trout (muscle biopsy) | Beef (muscle tissue biopsy) |
| Biological fluids | |
| chicken blood (in storage buffer) | |
Table 2:Animal sample types successfully tested using the sbeadex Lightning chemistry for DNA purification.
sbeadex Lightning has been successfully tested against popular plant DNA purification kits from market-leading competitors, showing comparable or even better DNA purities and yields (table 3) as well as a clear reduction of protocol steps and time – in addition to significant savings regarding consumption of plastic consumables and amount of liquid waste (table 4). Moreover, DNA isolated using sbeadex Lightning also showed a very good integrity compared to DNA isolated using those competitor kits (figure 4).
Please find a complete list of already tested plant and animal samples with a recommendation of the appropriate Core Kit and Lysis buffers/conditions here.
| Purification chemistry |
yield via fluorescence [µg DNA/ mg sample] |
yield via UV-Vis [µg DNA /mg sample] |
A260/280 | A260/230 |
|---|---|---|---|---|
| sbeadex Lightning (upscaled) | 0.5 | 0.9 | 1.8 | 1.5 |
| sbeadex maxi plant (upscaled) | 0.3 | 0.6 | 1.9 | 1.4 |
| Competitor A | 0.03 | 0.3 | 1.9 | 0.1 |
| Competitor B | 0.2 | 1.1 | 1.8 | 2.0 |
| Competitor C | 0.02 | 0.1 | 1.6 | 0.4 |
| Competitor D | 0.3 | 0.5 | 1.9 | 1.8 |
| Competitor E | 0.4 | 0.6 | 1.7 | 0.7 |
| Competitor F | 0.1 | 0.1 | 1.8 | 2.3 |
Table 3: A summary of DNA quantity and purity values for sbeadex Lightning, original sbeadex, and market-leading competitors. Values are based on processing of 15 mg maize leaf tissue samples. Both the sbeadex Lightning protocol and the sbeadex maxi plant protocol were upscaled to 400 µL lysate input to account for the different lysate input amounts used by competitors.
| Purification chemistry | Protocol steps* | Mean protocol time per sample** | Mean plastic waste per sample | Liquid waste per sample |
|---|---|---|---|---|
| sbeadex Lightning (upscaled) | 3 | 5 min | 2 g | 1.8 mL |
| Competitor A | 5 | 20 min | 6 g | 1.8 mL |
| Competitor B | 6 | 45 min | 9 g | 3.4 mL |
| Competitor C | 7 | 24 min | 7 g | 2.6 mL |
| Competitor D | 5 | 26 min | 8 g | 3.4 mL |
| Competitor E | 8 | 34 min | 9 g | 3.4 mL |
| Competitor F | 8 | 29 min | 19 g | 2.1 mL |
Table 4: A summary of the key savings for sbeadex Lightning compared to original sbeadex protocols and market-leading competitors. This table summarises the savings in protocol time, protocol steps, plastic consumables, and liquid waste. Values are based on processing of 15 mg maize leaf tissue samples. The sbeadex Lightning protocol was upscaled to 400 µL lysate input to account for the different lysate input amounts used by competitors. *excluding lysis step ** manual protocol
Figure 4: Gel electrophoresis image of DNA purified from maize leaf with sbeadex Lightning and competitor kits (compare figure 2 and table 2). The 0.8 % agarose gel was run at 80 V for 45 minutes and subsequently stained with ethidium bromide. Per sample, 6 µL purified DNA was added to each lane. 1: sbeadex Lightning (upscaled), 2: Competitor D, λ: 100 ng lambda DNA, 3: Competitor C, 4: Competitor B, 5: Competitor E, 6: Competitor F, 7: Competitor A.
Chemistry format
The sbeadex Lightning chemistry comes as a modular system to enable you to compile an optimal kit tailored to your specific needs. We provide two versions of sbeadex Lightning core kits (Kit A or Kit B) that contains a sbeadex Lightning binding buffer (LP in Kit A and LU in Kit B), sbeadex particles suspension and elution buffer AMP. These core kits can be complemented with a range of individual components (e.g. lysis buffers, Protease K solution, debris capture beads) that can be purchased separately.
To help with optimisation of the lysis conditions, we also provide a sbeadex Lightning Starter Kit that includes the binding buffers of both core kits, six different lysis buffers (PN, PVP, UR, BL, H, LI), Protease K solution and debris capture beads. Our Quick guide for the sbeadex Lightning Starter Kit will help you to test and determine your optimal lysis and purification conditions.
How to get started with sbeadex Lightning technology

Order a sbeadex Lightning Starter Kit and test this with your samples. Once you have optimised your protocol, (2.) select/order the components to generate your tailor-made sbeadex Lightning solution (3.). *Please find sample-specific standard protocols online or contact techsupport@lgcgroup.com for assistance.
Note: Our first sbeadex Lightning protocols have been developed for purification of DNA from plant and animal samples but we are already developing protocols for other sample types. We have observed that sbeadex Lightning is also suitable for RNA isolation and are currently also validating this application.
If you are working with samples other than plant or animal, the sbeadex Lightning Nucleic Acid Purification Starter Kit can still be used to evaluate whether sbeadex Lightning is appropriate for your specific sample types.
We offer our Starter Kit, which includes 20 preparations, to help evaluate and optimise DNA purification workflow. Order a starter kit now!
Experience the evolution of nucleic acid purification
- High purity: Elevate your research with nucleic acids of exceptional purity, meticulously isolated using our advanced sbeadex magnetic bead technology.
- Streamlined protocols: Experience efficiency in every step. Our kits offer fast, convenient, and reproducible protocols that seamlessly integrate into high-throughput automation, saving you time and effort.
- Versatile applications: From basic and biopharmaceutical research to molecular diagnostics, our sbeadex technology is applicable across a diverse range of sample types, ensuring adaptability to your specific research needs.
- Inhibitor-free: Bid farewell to potential PCR inhibitors. Our water-based wash buffers are meticulously designed to eliminate any hindrance to your downstream applications, providing nucleic acids ready for various assays.
- Stability and compatibility: Trust in the high stability of our ready-to-use kits, compatible with popular robotic platforms such as KingFisher, Hamilton, Tecan, Beckman-Coulter, or our high-throughput oKtopure instrument. Your research deserves reliable tools, and we deliver.
Harness the power of sbeadex technology
Quality is in our DNA. Our mission is to deliver the highest quality products and services and consistently exceed customer expectations. We continue to make investments to ensure that our design, manufacturing and shipping processes comply with industry standards.
In contrast to traditional methods, our technology streamlines the process by eliminating the need for a drying step before elution, ensuring exceptional DNA quality. Prioritising sustainability, our kits feature an ethanol-free second wash buffer which helps to remove impurities that might inhibit enzymatic downstream reactions.
Versatility is at the core of our technology, making it suitable for diverse applications, from basic biopharmaceutical research to molecular diagnostics, and agricultural screening. The adaptable and reproducible protocols, along with compatibility with popular robotic platforms, position sbeadex kits as a reliable solution for high-throughput automation.
Efficient DNA/RNA preparation with sbeadex magnetic beads: streamlined protocol for high-quality nucleic acids
Discover a rapid and efficient DNA/RNA preparation protocol using sbeadex magnetic beads. Achieve high-quality purification of small and large DNA and RNA fragments within just 45 minutes, eliminating the need for centrifugation steps. Our double-coated beads employ a unique dual binding protocol, enabling the elution of ultrapure nucleic acids directly into water. Experience reduced solvent carryover, ensuring the integrity of your samples for optimal downstream applications
sbeadex plant kits have been successfully used with:
| Plant species | Leaves | Seed |
|---|---|---|
| Apricot (Prunus armeniaca) | X | |
| Barley (Hordeum vulgare) | X | X |
| Beet, sugar (Beta vulgaris) | X | |
| Canola/oilseed (Brassica napus) | X | X |
| Chicory (Cichorium intybus) | X | |
| Corn (Zea mays) | X | X |
| Cotton (Gossypium) | X | X |
| Cucumber (Cucumis sativus) | X | X |
| Flax (Linum usitatissimum) | X | |
| Grape (Vitis vinifera) | X | X |
| Lettuce (Lactuca sativa) | X | |
| Muskmelon (Cucumis melo) | X | X |
| Onion (Allium cepa) | X | X |
| Parsley (Petroselinum crispum) | X | X |
| Peach (Prunus persica) | X | |
| Pepper (Capsicum annuum) | X | X |
| Potato (Solanum tuberosum) | X | |
| Rice, Asian (Oryza sativa) | X | X |
| Rubber (Hevea brasiliensis) | X | X |
| Soybean (Aphis glycines) | X | X |
| Sunflower (Helianthus annuus) | X | X |
| Tobacco leaves (Nicotiana tabacum) | X | X |
| Tomato (Solanum lycopersicum) | X | X |
| Wheat (Triticum L.) | X | X |
Typical quantities of starting material are:
- 5-10 mg lyophilised tissue or 10-30 mg fresh tissue for the sbeadex Mini Plant DNA Purification Kit
- 10-30 mg lyophilised tissue or 40-120 mg fresh tissue for the sbeadex Maxi Plant DNA Purification Kit
sbeadex plant DNA purification resources
| Title | Type |
|---|---|
| General sbeadex Plant DNA Purification Kit manual (valid for sbeadex Mini and Maxi kits) | Manual |
| sbeadex Mini Plant DNA Purification Kit (trial kit) | Protocol |
| sbeadex Mini Plant DNA Purification Kit | Protocol |
| sbeadex Maxi Plant DNA Purification Kit | Protocol |
| sbeadex Plant DNA Purification Kits | Data sheet |
| sbeadex plant kit application note (for seed) | Application note |
| The oKtopure™ and sbeadex™ plant nucleic acid extraction kit | Application note |
| sbeadex maxi plant kit and KingFisher 96 instrument | Application note |
| sbeadex mini plant kit and KingFisher 96 instrument | Application note |
| oKtopure automated nucleic acid purification | Data sheet |
| sbeadex plant DNA purification safety data sheets | SDS |
The sbeadex Pathogen testing kits have been used successfully with:
| sbeadex Pathogen Nucleic Acid Purification Kit | Competitor A | ||||
| N | Average Cq | StDev | Average Cq | StDev | |
| Clean up sample | 12 | 30.35 | 0.14 | 33.80 | 0.15 |
| Direct spike-in sample | 3 | 31.00 | 0.44 | 33.80 | 0.12 |
Figure 1: Demonstration of no loss of RNA during the sbeadex Pathogen Nucleic Acid Purification Kit protocol. 500 copies of a 2019-COVID artificial RNA reference material were added to a sample (nasopharyngeal swab) and the sample processed with the sbeadex Pathogen Nucleic Acid Purification Kit. SARS-CoV-2 was detected in this preparation and a directly spiked positive control sample via a RT-qPCR at two separate Biosearch Technologies sites. The concordant Cq values from both samples demonstrate no loss of sample due to the extraction step.
| Pathogen | External Control (Artificial Matrix) |
|---|---|
| Viral Targets | |
| SARS-CoV-2 | VTM1 |
| Influenza A H3N2 | Swab2 |
| Influenza B | Swab2 |
| Novel Influenza A H1N1 | Swab2 |
| Respiratory Syncytial Virus (subtype A) | Swab2 |
| Respiratory Syncytial Virus (subtype B) | Swab2 |
| Adenovirus 4 | Swab2 |
| Zika Virus | Plasma3 |
| Cytomegalovirus | Plasma4 |
| Human Papillomavirus-16 | Methanol-preserved cultured human cells containing full-length HPV episomal DNA5 |
| Bacterial Targets | |
| Bordetella pertussis | Swab6 |
| Chlamydophila pneumoniae | Swab6 |
| Chlamydophila psittaci | Swab6 |
| Coxiella burnetii | Swab6 |
| Legionella pneumophila | Swab6 |
| Mycoplasma pneumoniae | Swab6 |
| Mycobacterium tuberculosis | Sputum7 |
| Campylobacter jejuni | Stool8 |
| Salmonella enteritidis | Stool8 |
| Yersinia enterocolitica | Stool8 |
| Clostridium difficile | Stool8 |
| Chlamydia trachomatis | Urine9 |
| Neisseria gonorrhoeae | Urine9 |
| Staphylococcus aureus | External Run Control10 |
| Other Targets | |
| Candida albicans | Positive Extraction Control11 |
| Compatible Matrices Tested | Swabs (UTM®/VTM) Sputum Saliva Blood Plasma Serum Urine Stool Cerebrospinal Fluid |
Table 1: Pathogens that have already been tested using the sbeadex Pathogen Nucleic Acid Purification Kit.
Standards used for testing
1. AccuPlex™ SARS-CoV-2 Verification Panel - Full Genome, 0505-0168 (LGC SeraCare)
2. AMPLIRUN® TOTAL RESPIRATORY VIRAL PANEL CONTROL (SWAB), MBTC020 (Vircell S.L.)
3. AMPLIRUN TOTAL ZIKV/DENV/CHIKV CONTROL (PLASMA), MBTC023 (Vircell S.L.)
4. AMPLIRUN TOTAL CMV CONTROL (PLASMA), MBTC018-R (Vircell S.L.)
5. AccuTrak™ HPV Genotype Qualification Panel, QSH701 (2400-0161), (LGC SeraCare)
6. AMPLIRUN TOTAL ATYPICAL BACTERIAL PNEUMONIA CONTROL (SWAB) MBTC022-R (Vircell S.L.)
7. AMPLIRUN TOTAL MTB CONTROL (SPUTUM) MBTC013 (Vircell S.L.)
8. AMPLIRUN TOTAL GASTROINTESTINAL BACTERIAL PANEL CONTROL (STOOL), MBTC021 (Vircell S.L.)
9. AMPLIRUN TOTAL CT/NG CONTROL (URINE), MBTC003 (Vircell S.L.)
10. NATtrol™ S. aureus (MRSA) External Run Control, NATSAU(MRSA)-ERCM (Zeptometrix Corporation)
11. NATtrol Candida/TV Positive Control, NATCTVPOS-BD (Zeptometrix Corporation)
Note: Other sample matrices may also be used with this kit. Further testing on other pathogens and matrices is in progress and this table will be updated accordingly.
sbeadex pathogen purification resources
The sbeadex livestock DNA testing kits have been used successfully with:
| Sample type | Animal species | |
|---|---|---|
|
|
|
sbeadex livestock DNA purification resources
| Title | Type |
|---|---|
| sbeadex Livestock DNA Purification Kit | Manual |
| sbeadex Livestock DNA Purification Kit | Data sheet |
| oKtopure automated nucleic acid purification | Data sheet |
| oKtopure tip wash station | Technical note |
| Genotyping solutions | Flyer |
| sbeadex livestock DNA purification safety data sheets | SDS |
sbeadex forensic purification resources
| Title | Type |
|---|---|
| sbeadex Forensic DNA Purification Kit | Manual |
| sbeadex Forensic DNA Purification Kit | Data sheet |
| sbeadex Forensic DNA Purification Kit for automated processing | Application note |
| sbeadex Forensic PCR clean up | Application note |
| Sensitivity of the sbeadex Forensic DNA Purification Kit | Application note |
| Validation of custom sbeadex Forensic DNA Purification Kit | Application note |
| Comparison of the sbeadex Forensic DNA Purification Kit | Application note |
| sbeadex Forensic DNA Purification Kit and the KingFisher 96 instrument | Application note |
| sbeadex forensic DNA purification safety data sheets | SDS |
sbeadex blood DNA purification resources
| Title | Type |
|---|---|
| sbeadex Blood DNA Purification Kit | Manual |
| sbeadex Blood DNA Purification Kit | Data sheet |
| sbeadex Blood DNA Purification Kit and anticoagulant preservatives | Application note |
| Automation of the sbeadex Blood DNA Purification Kit using Hamilton robotics | Application note |
| sbeadex blood DNA purification safety data sheets | SDS |
sbeadex plasmid DNA purification resources
| Title | Type |
|---|---|
| sbeadex Plasmid DNA Purification Kit | Manual |
| sbeadex Plasmid DNA Purification Kit and KingFisher 96 instrument | Protocol |
| sbeadex plasmid DNA purification safety data sheets | SDS |
sbeadex Tissue DNA Purification Kits have been successfully used with:
|
sbeadex tissue DNA purification resources
sbeadex PCR clean-up kit resources
| Title | Type |
|---|---|
| sbeadex PCR clean-up Kit | Manual |
| sbeadex forensic PCR clean-up | Application note |
| sbeadex forensic PCR clean-up safety data sheets | SDS |