Choice of Quenchers: The evolution from TAMRA to Dabcyl to BHQ labels
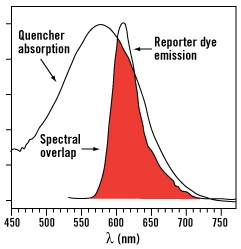
FAM-TAMRA probes, oligonucleotide probes with fluorescein (FAM) as a 5' label and tetramethylrhodamine (TAMRA) as a 3' label, were commonly used before the availability of dark quenchers and are still used by some groups today. With these probes, FAM is the reporter dye and TAMRA is the acceptor dye. These probes work well due to the good spectral overlap between the FAM fluorescence and TAMRA absorption curves. In a FRET assay, the FAM dye is excited at 490 nm and transfers energy to TAMRA. The FRET mechanism returns the FAM reporter to the ground state and generates TAMRA in an electronically excited state without any light emission or absorption. Due to FRET, the amount of FAM fluorescence loss at 520 nm is proportional to the amount of TAMRA fluorescence gain at 580 nm.
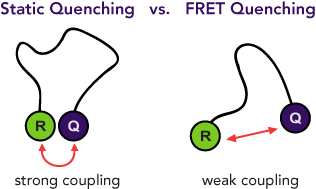
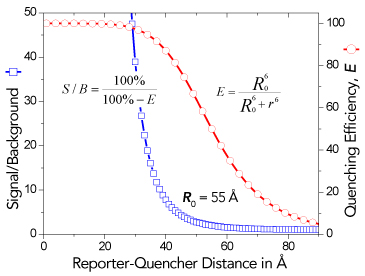
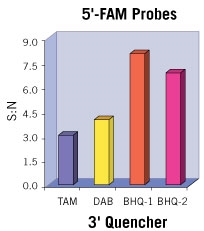
Unfortunately, when a fluorescent dye, such as TAMRA, is used as a quencher, its fluorescence can contribute to background signal. In addition, such quencher fluorescence makes multiplexing, in which more than one reporter-quencher pair is used simultaneously in a reaction, more difficult. These limitations led to the use of Dabcyl which is known as a dark quencher because it is not fluorescent. Dabcyl, however, has limited utility as a FRET quencher because it has an absorption maximum at 453 nm, which is blue-shifted and far removed from the emission maxima of most reporters. Therefore, Dabcyl has poor spectral overlap and consequently minimal FRET quenching.
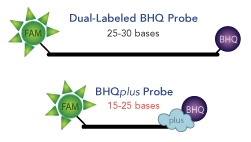
Black Hole Quencher® (BHQ®) labels are a family of dark quenchers with excellent spectral overlap with all common reporter dyes for efficient FRET quenching. As a result, BHQ labels have become the industry standard for quenchers in dual-labeled hydrolysis probes and many other molecular probe formats.
The most recent or “highly evolved” addition is the BHQplus™ probe. The BHQplus probe is a novel, compact probe for qPCR with a duplex stabilizing technology terminated with Biosearch’s Black Hole Quencher dye. The stabilizing technology permits the design of shorter oligonucleotides which enables greater specificity as well as the detection of more difficult targets such as AT-rich regions. BHQplus probes are especially well suited to detect single nucleotide polymorphyisms.