Design exactly what you need with our
expansive menu of options.
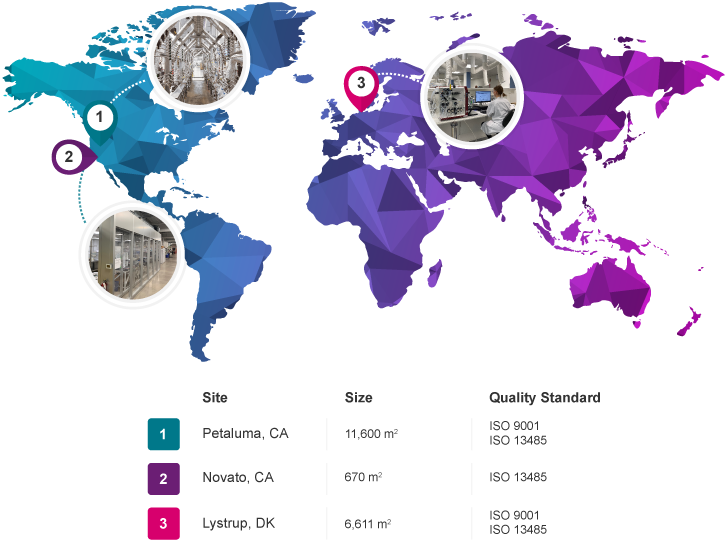
Leverage our multi-site oligonucleotide manufacturing operations to produce oligos with the quality, reliability and scalability you need, from research grade to commercial diagnostics and therapeutic applications.
Quality from research grade to commercial use
Quality is in our DNA. Our mission is to deliver the highest quality products and services and consistently exceed customer expectations. We continue to make investments to ensure that our design, manufacturing and shipping processes comply with industry standards.
Biosearch Technologies uses the ISO family of internationally recognized Quality Management System standards as the framework for organizing, evaluating and improving our work. Compliance with these standards is assessed through extensive regular audits performed by qualified registrar agencies. Successful audits lead to certification of the company site’s management system to the applicable standard, valid for three-year intervals.
Evaluate choices for probe selection and design
Learn more about the advantages of each probe type based and explore resources to support selecting the best probe chemistry for your experimental logistics.
Educate yourself on everything oligos
- Educate yourself on everything oligos
- Understand the nuances of probe selection and design with this eBook
- Wondering about the use of MGB, LNA and BHQplus? Read this article
- Use the probe technology selector tool
- Find the right dye to maximise results using the spectral overlay tool
- Evaluate oligo specifications and perform common assay calculations